I would say that the these two ideas are not in opposition, but rather are complementary, in fact ideally one addresses a situation when the other has completed it's function. To use only one option would be an inefficient use of the available possibilities.
I first came across this confusion while in New Delhi, India, as I was working on some water harvesting projects there. I was presented to someone who was introduced to me as an expert on "Rainwater Harvesting", he had a PhD, and over 30 years of experience, working with the national government of the topic. We started talking and agreed that the best thing to for water management was "harvesting", it was the most practical and cost effective means of obtaining water. However when I asked him how he went about doing it, he described to me a dry well system for aquifer recharge.
I asked him, "Okay, that's good what you do with the overflow, but what about how you harvest it?", the answers was "Yes, this is how we are harvesting rainwater."
Evidently there was a misunderstanding, we were talking about 2 different things, though related in some way, and the same name was being used for both.
The use of correct names is very important, many times we find that because names and designation have been used incorrectly then problems occur. There is a further and added complication that language or languages are dynamic entities, as technology and science evolve, new words get invented to name new things.
Not so long ago I started and created the Wikipedia article for Stormwater Harvesting, differentiating it from Rainwater Harvesting. This is a consequence of advance in technology and refinement of techniques.
Apart from this discussion of "name calling", we should be addressing what is important. I would say that the most important objective to be achieved is to have an implementation that is the most cost effective, practical and simple, to make high quality water immediately available for use with minimum, if any, power consumption. Ideally it should also be done in the most sustainable way possible.
If this is the objective, then all we have to do is find the way of achieving this objective.
To date, the most cost effective way to store water indefinitely and not only conserve it's quality, but also improve it, are in covered underground systems, that offer high surface are to volume ratios such as I have described in a previous article, where the positive of effects biomass are explained. Above ground tanks might be cheaper, however they are poor performing in terms of water quality conservation. Open water dams have problems of high level of evaporation, as well as problems with vermin and water quality degradation, from various other sources.
Someone might say though that, the ability to store water is something limited, and this is correct, it is then in this case that we should look also at aquifer or ground water recharge. Once the capacity of the underground water system has been filled then the overflow can be used in a very effective infiltration method, to recharge the water table. This method then not only has the best of both worlds, having water for immediate reuse, but also recharging the aquifer, however not only that, but if the overflow water has passed through a suitable system it is purified and goes into the aquifer it is also better for the health of the aquifer.
In recent time aquifers have suffered substantially in different aspects. It is important to replenish aquifers. Only recently have people come to realize the water is a finite resource that has to managed responsibly and with much care. It can be said that this is a similar situation to what happened with oil, what was originally though to be a resource that would last forever, became something very precious.
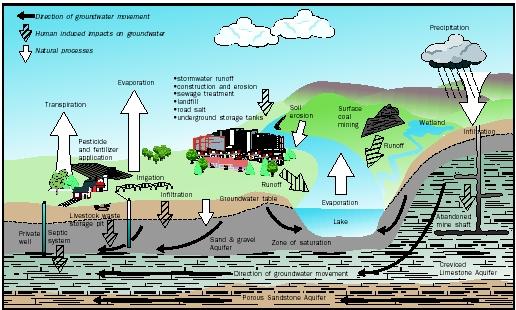
In the drawing below we illustrate how modern urbanisation has reduced the ability of rain or stormwater to infiltrate and enter the aquifers, not only that, but the water quality of what little infiltration occurs, is probably not very good for the aquifer as it has picked up a lot of surface contaminants. "Modern" techniques of dry wells, though give the impression of creating some good, in terms of replenishing aquifers can be more harmful than good, this is especially true in industrial areas where discharge of toxic chemicals can also enter the aquifer.
 |
| source: http://www.landscapeforlife.org/water/3b.php |
 |
| source: http://www.waterencyclopedia.com/ |
In some places such as India, this happens at an alarming rate:
The groundwater table is Delhi has depleted to 20 –30 metres in various areas across the city. Compared to a level of 30 – 40 feet at the time of Independence, the water table has dropped to 350 feet at certain places. It is said to be falling at 10 feet per year on an average. Groundwater levels have depleted by 2 – 6m in Alipur and Kanjhwla blocks, 10m ins the Najafgarh block, and about 20 m in Mehrauli block.Some are even more dramatic in their statements, an article from the Harvard Business School Alumni Bulletin states:
source: http://www.rainwaterharvesting.org/index_files/about_delhi.htm
"groundwater supplies in Delhi are expected to run dry by 2015"
source: http://hbswk.hbs.edu/archive/5049.html
Another aspect of concern is called seawater intrusion, this happens in more coastal areas. Significant studies have been conducted into this phenomenon:
Seawater intrusion in coastal aquifers by Food and Agriculture Organization of the United Nations. Land and Water Development Division
In relation to aquifer contamination an interesting paper is An Overview of the Current Research on Remediation of Soils and Aquifers Contaminated by Organic Compounds by C.D. Johnston and G.B. Davis.
In the figure below we can see that groundwater contamination is not something to be dealt lightly with, in fact the situation that the areas that are most vulnerable are agricultural areas indicate a very noxious cycle.
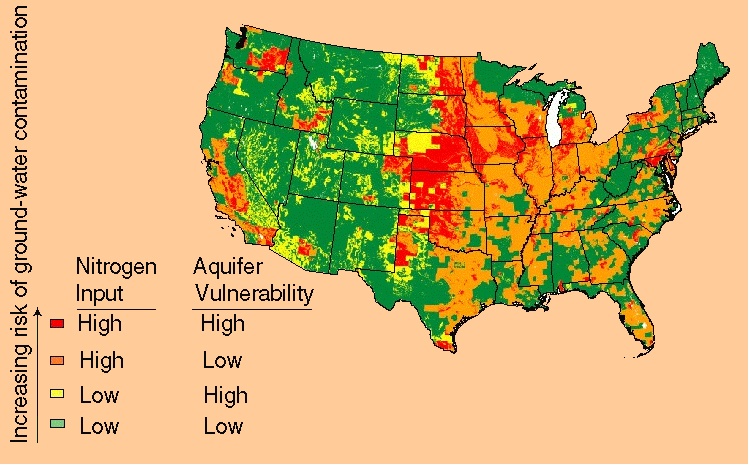 |
| source: http://water.usgs.gov |
So what is the solution? There are various, but from a consumer point of view it something that is not that complicated. In the diagram below is a simple representation of how a combined rainwater harvesting and overflow infiltration system is configured. The combination of underground tanks used to store water for immediate reuse, as well as to allow a separate tank for infiltration, that at the same time purifies the water, that is maintenance free, that requires minimum amount of power to pump the water for reuse as is practically just below the surface. Pumps for elevating and pumping ground water must be much more powerful, are as a consequence much more expensive, and in areas with rapidly descending water tables, this becomes a costly element if one has to the change the pump every few years to cope with the lowering water table.
Above we can see that with modular tanks, there is design flexibility, in relation to the amount of storage capacity for immediate reuse, as well as the infiltration tank. This way a project can harvest as much water as is necessary, conserve it ideal conditions. Water that exceeds the capacity of the tank for immediate reuse goes into the infiltration tank, and does so at an exceptional quality, and can go safely back into the aquifer.